Antimicrobial Resistance:
The Fight to Preserve Our Last Line of Defense
Uncovering the Challenges, Confronting Misguided Therapies, and Driving Strategic Action to Protect Treatable Infections
How does antimicrobial resistance (AMR) develop?
Antimicrobials is the term used to describe all medicines that treat and prevent infections in humans, animals and plants.1 They include antibiotics, antivirals, antiparasitics, and antifungals.1 Over time, microorganisms undergo changes that mean they no longer respond to antimicrobials.2 These treatment-resistant pathogens grow and spread, making infections difficult or impossible to treat and leading to an increased risk of diseasespread.2
AMR burden
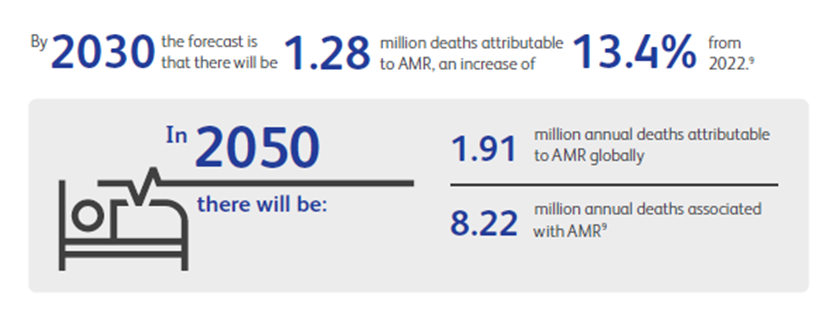
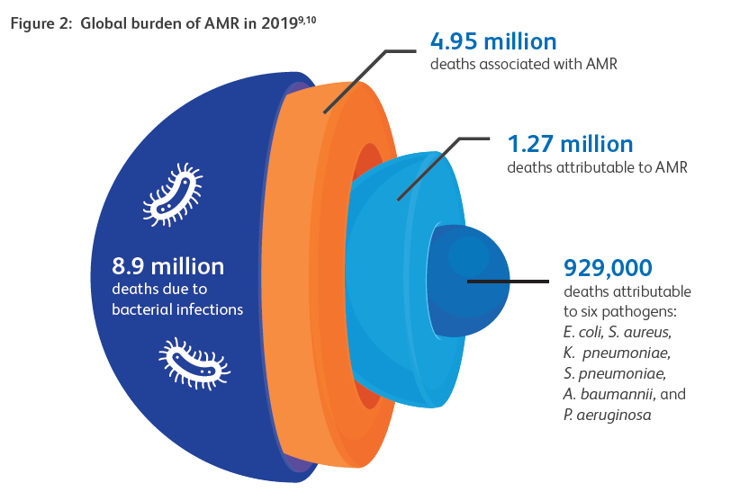
In 2021, approximately 5 million deaths were associated with AMR, including cases involving drug-resistant lower respiratory tract and bloodstream infections3,4.
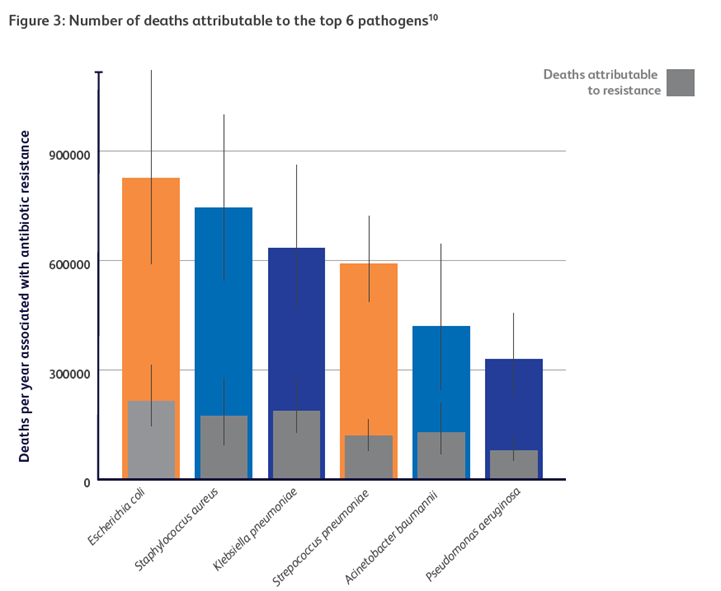
Currently, more than 1 million deaths occur annually as a direct consequence of bacterial resistance to antimicrobial therapy3,4.
Estimated annual costs (in US$) and benefits in 2050 of better treatment for bacterial infections and innovative new gram-negative drugs6
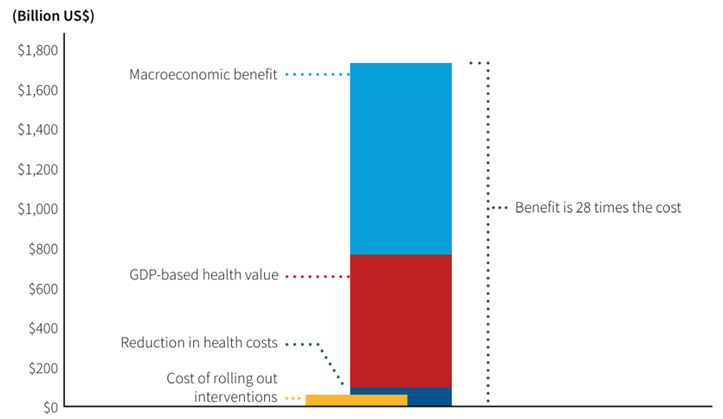
AMR is associated with substantial increases in healthcare costs5,6
The current direct health care costs associated with AMR are US$ 66 billion per year (0.7% of global health expenditures).5
Estimates project that globally, AMR may result in:
• US$1 trillion in additional healthcare costs by 2050
• US$1 trillion to US$3.4 trillion gross GDP losses/year by 2030
Learn More and Download our AMR Booklet

A treatable infection doesn’t harm the patient – misguided therapy does
When resistant organisms are falsely reported as susceptible – a laboratory error known as a very major error (VME), clinicians can be misled, resulting in ineffective treatment Antimicrobial therapies guided by inaccurate AST results generated by certain automated systems were associated with a 2x increase in mortality rates7
What if you could get better, more accurate results?

VMEs can critically mislead clinical decisions and delay life-saving treatment.
VMEs are associated with inappropriate antimicrobial therapy decisions and poorer outcomes, allowing infections to progress unchecked, potentially leading to sepsis, organ failure, death, and contributing to growing antimicrobial resistance7,8
Are you treating with confidence or compromise?
Download now our AMR Educational Booklet and stay ahead in the fight against AMR
This resource uncovers the critical challenges driving AMR and outlines key strategies for action, from robust surveillance systems to integrated approaches across human, animal, and environmental health. Equip yourself with the latest insights and practical solutions - because together, we can safeguard health and preserve life-saving treatments.
Treat with confidence
BD Phoenix™ M50 offers enhanced VME performance over competing systems
BD Phoenix™ M50 demonstrates lower rate of VMEs for Gram-Negative organisms9,10.

NEW PANELS: Because Every Well Counts
BD Phoenix™ Panels Deliver True MICs and Complete Breakpoint Coverage for Unmatched Accuracy11,12
Optimized well layout
Accommodates all clinically relevant antimicrobials reducing the need for offline testing
Future-ready flexibility
Layout allows for updates as resistance patterns and guidelines evolve

The new BD Phoenix™ panels include the latest dilution ranges aligned with EUCAST breakpoints, updated according to the most recent recommendations, and now feature the newest concentration for Trimethoprin/sulfamethoxazole (SXT) to ensure accurate detection of resistance at or near decision thresholds13.
- Incidence of Carbapenemase-producing CPE (CP-CRE) increased 69%11
- NDM-type CPE, New Delhi metallo-β-lactamase surged 461%11
- NDM and OXA-48 Carbapenemases are rising10
- NDM was found in 27% of carbapenem-resistant E. coli, 24% of Klebsiella spp, and 6% of Enterobacter spp.11

BD Phoenix™ – effective workflow to minimize offline testing
BD Phoenix™ is designed to avoid downstream inefficiencies, the 136-well design provides the highest analytical throughput per panel enabling inclusion of all relevant antimicrobials, minimizing offline testing12,14

Less repetitive manual tasks to focus on what matter most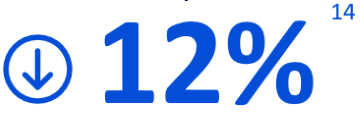
BD Phoenix™ AP automates McFarland preparation reducing hands-on time by 12% compared to bioMerieux Vitek®14
BD Phoenix™ and BD Synapsys™: Your Stronger Defense Against AMR
The BD Bloodstream Infection Solution with BD Phoenix™ and the dynamic intelligence of BD Synapsys™ is helping microbiology labs to strengthen your defense against antimicrobial resistance Register here to stay up to date on BD Synapsys ™ and BD Phoenix ™
Register here to stay up to date on BD Synapsys ™ and BD Phoenix ™
Register here to stay up to date with BD Synapsys™ and BD Phoenix™